Interstitial flow differentially increases patient-derived glioblastoma stem cell invasion via CXCR4, CXCL12, and CD44-mediated mechanisms†
Abstract
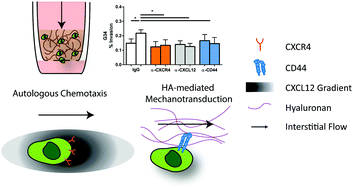
Glioblastoma (GBM) prognosis remains dismal due in part to the invasiveness of GBM cells. Interstitial fluid flow (IFF) has been shown to increase invasion of glioma cells in vitro through the CXCR4 receptor interacting with autologous, pericellular gradients of CXCL12 (autologous chemotaxis) or through the CD44 receptor interactions with the extracellular matrix (hyaluronan-mediated mechanotransduction). These mechanisms have not been examined together and thus we hypothesized that both mechanisms contribute to invasion in populations of cancer cells. Therefore, we examined IFF-stimulated CXCR4-, CXCL12-, and CD44-dependent invasion in patient-derived glioblastoma stem cells (GSCs). Using our 3D in vitro assay and correlative in vivo studies we demonstrated GSC lines show increased invasion with flow. This flow-stimulated invasion was reduced by blockade of CXCR4, CXCL12, and/or CD44, revealing that GSC invasion may be mediated simultaneously by both mechanisms. Characterization of CXCR4+, CXCL12+, and CD44+ populations in four GSC lines revealed different percentages of protein positive subpopulations for each line. We developed an agent-based model to identify the contributions of each subpopulation to flow-stimulated invasion and validated the model through comparisons with experimental blocking studies. Clinically relevant radiation therapy increased flow-stimulated invasion in one GSC line. Our agent-based model predicted that IFF-stimulated invasion is driven primarily by CXCR4+CXCL12+ populations, and, indeed our irradiated cells had an increase in this subpopulation. Together, these data indicate that different mechanisms govern the flow response across GSCs, but that within a single patient, there are subpopulations of GSCs that respond to flow via either CD44- or CXCR4–CXCL12 mechanisms.

 Please wait while we load your content...
Please wait while we load your content...