Future perspectives for formaldehyde: pathways for reductive synthesis and energy storage
Abstract
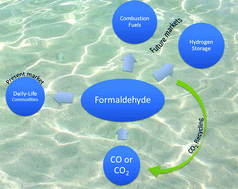
Formaldehyde has been a key platform reagent in the chemical industry for many decades in a large number of bulk scale industrial processes. Thus, the annual global demand reached 30 megatons per year, and currently it is solely produced under oxidative, energy intensive conditions, using high-temperature approaches for the methanol oxidation. In recent years, new fields of application beyond the use of formaldehyde and its derivatives as i.e. a synthetic reagent or disinfectant have been suggested. For example dialkoxymethane could be envisioned as a direct fuel for combustion engines or aqueous formaldehyde and paraformaldehyde may act as a liquid organic hydrogen carrier molecule (LOHC) for hydrogen generation to be used for hydrogen fuel cells. To turn these new perspectives in feasible approaches, it requires also new less energy-intensive technologies for the synthesis of formaldehyde. This perspective article spreads light on the recent directions towards the low-temperature reductive synthesis of formaldehyde and its derivatives and low-temperature formaldehyde reforming for hydrogen generation. These aspects are important for the future demands on modern societies’ renewable energy management, in the form of a methanol and hydrogen economy, and the required formaldehyde-feedstock for the manufacture of many formaldehyde-based daily products.
- This article is part of the themed collection: Harvesting Renewable Energy with Chemistry


 Please wait while we load your content...
Please wait while we load your content...