An enolate ion as a synthon in biocatalytic synthesis of 3,4-dihydro-2(1H)-quinoxalinones and 3,4-dihydro-1,4-benzoxazin-2-ones: lemon juice as an alternative to hazardous solvents and catalysts†
Abstract
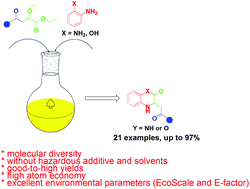
Innovative, efficient, clean, experimentally simple and environmentally friendly one-pot biocatalytic synthesis of two small libraries of 3,4-dihydro-2(1H)-quinoxalinones (4a–m) and 3,4-dihydro-1,4-benzoxazin-2-ones (5a, 5b, 5f and 5k–o) by heterocyclization of ethyl 2-hydroxy-4-alkyl(aryl)-4-oxo-2-butenoates (1a–o) or their corresponding salts (1a′–o′) with o-phenylenediamine (2) or o-aminophenol (3) in lemon juice as a solvent and a catalyst is presented. In all reactions where esters 1a–o are used for 24 h, very good-to-excellent yields were achieved, but the best yield was realized in the synthesis of 5a (97%) from 3 and 1a. The use of enolate salts (1a′–o′) instead of the corresponding esters 1a–o significantly reduced the reaction time (up to 6 h) and good-to-excellent yields were achieved. Groups with electron-donating or -withdrawing effects at the aromatic ring of the ester did not have significant influences on the yield of targeted products. In addition, several selected 3,4-dihydro-2(1H)-quinoxalinones and 3,4-dihydro-1,4-benzoxazin-2-ones on a gram-scale in the yields up to 92% were synthesized. The presented synthetic strategy has produced smaller amount of waste without by-products and with excellent values of green chemistry metrics (atom efficiency, reaction mass efficiency, E-factor and EcoScale).

 Please wait while we load your content...
Please wait while we load your content...