Nitrogen oxides reduction by carbon monoxide over semi-coke supported catalysts in a simulated rotary reactor: reaction performance under dry conditions†
Abstract
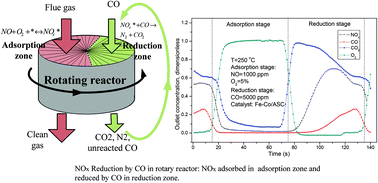
A novel rotary reactor was proposed for nitrogen oxides (NOx) reduction by carbon monoxide (CO) in order to avoid the oxygen inhibition effect. NOx adsorption and reduction were separated into two different zones in the proposed rotary reactor. NOx in the flue gas was adsorbed onto the catalyst surface in the adsorption zone and then reduced by the reducing gas in the reduction zone. The performance of the proposed reaction process was investigated in a simulated rotary reactor. The catalysts used were lab synthesized, Fe and Co co-impregnated over activated semi-coke. The results showed an excellent NOx removal efficiency at 200 and 250 °C. However, the NOx reduction efficiencies were always much lower than the removal efficiency. Most of the removed NOx was simply passed from the flue gas to the reducing gas. It is very important to feed a great excess of CO in the reduction zone, since CO tended to be firstly consumed by residual oxygen in the reduction zone and then be involved in the NO + CO reaction. In an in situ DRIFT study on the dynamic NOx adsorption–reduction process, it was found that poor reduction and desorption behavior of the stored nitrates was the bottleneck of the NOx adsorption–reduction process.

 Please wait while we load your content...
Please wait while we load your content...