Selective conversion of castor oil derived ricinoleic acid methyl ester into jet fuel
Abstract
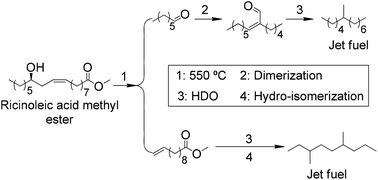
The selective conversion of ricinoleic acid methyl ester into jet fuel was realized through the integration of feedstock properties and rational catalytic design strategy. A unique hydroxyl group in the ricinoleic acid chain induced a special thermal rearrangement reaction in medium chain fatty acid methyl ester (FAME) and heptanal formation. Such a reaction was used as the starting point for our proposed process. Methyl 10-undecenoate was converted into a branched paraffin with limited carbon loss. Both Pt/Al2O3 and Pt/ZSM-22 catalysts resulted in selective hydrogenation (with limited decarbonylation) and hydro-isomerization (with negligible cracking), respectively. Another product, heptanal, was also converted into jet fuel through selective dimerization and hydrodeoxygenation with a supported amine and commercially available Pt/Al2O3 catalyst, respectively. With the right integration of these technologies, the carbon selectivity in the castor oil to jet fuel process is as high as 90%.

 Please wait while we load your content...
Please wait while we load your content...