Electrochemical C–H functionalization and subsequent C–S and C–N bond formation: paired electrosynthesis of 3-amino-2-thiocyanato-α,β-unsaturated carbonyl derivatives mediated by bromide ions†
Abstract
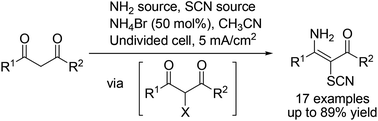
An efficient paired electrosynthesis involving C–H functionalization and subsequent C–S and C–N bond formation for the assembly of valuable 3-amino-2-thiocyanato-α,β-unsaturated carbonyl derivatives has been developed. In the paired electrolysis, the amino and thiocyanato moieties originate from a single reagent or a combination of ammonium acetate and potassium isocyanate. The chemistry proceeds in a simple undivided cell employing a sub-stoichiometric amount of NH4Br that serves both as an inner sphere type redox catalyst and a supporting electrolyte; in this manner an additional conducting salt is not required. The reaction also works using a catalytic amount of NH4Br. Cyclic voltammetry and the results of control experiments demonstrate that the reaction proceeds via an anodically initiated C–H functionalization of the 1,3-dicarbonyl substrates that occurs via the electrochemical oxidation of bromide and simultaneous cathodic reduction of ammonium ions.

 Please wait while we load your content...
Please wait while we load your content...