Efficient formation of nitriles in the vapor-phase catalytic dehydration of aldoximes
Abstract
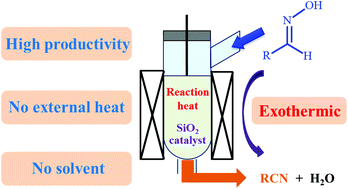
A vapor-phase dehydration of acetaldoxime to acetonitrile was investigated over various solid catalysts. Among the tested catalysts, ZrO2, Al2O3 and SiO2 showed high catalytic activity for the formation of acetonitrile from acetaldoxime, while the correlation between catalytic activity and the acid property of the catalysts was not observed. Weak acidic sites such as silanols sufficiently work as catalytic sites for the dehydration, which does not require strong acids such as zeolites. Several SiO2 catalysts with different physical properties were tested, and the SiO2 with the smallest pore size and the highest specific surface area showed the highest catalytic activity for the formation of acetonitrile. Because the dehydration of acetaldoxime to acetonitrile is exothermic, a large amount of reaction heat was generated during the reaction, and the reaction temperature was found to be significantly affected by the feed rate of the reactant and the flow rate of the carrier gas. In order to effectively utilize the in situ generated reaction heat, the dehydration of acetaldoxime to acetonitrile without using the external heat supply was conducted. The temperature was controllable even in the absence of the external heat, and the acetonitrile yield higher than 90% could be achieved in such a green operation under the environment-friendly adiabatic conditions.

 Please wait while we load your content...
Please wait while we load your content...