Hydrogenative cyclization of levulinic acid into γ-valerolactone by photocatalytic intermolecular hydrogen transfer†
Abstract
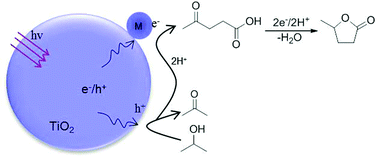
The hydrogenative cyclization of levulinic acid (LA) into γ-valerolactone (GVL) is an attractive route toward the use of renewable bio-sources but it normally suffers from the consumption of H2. In this study, we report that an intermolecular hydrogen transfer from isopropanol to LA can be realized efficiently under photocatalytic conditions over gold-loaded TiO2 catalysts. In this manner, isopropanol is dehydrogenated as acetone and pinacol with the total selectivity of >99%, whereas LA is hydrogenated and cyclized as GVL with the selectivity of up to 85%. In this reaction process, the production of GVL is mediated with hydrogenated dehydration of LA into an acetyl propionyl radical, which is further hydrogenated and cyclized as GVL. This hydrogenation–dehydrogenation coupling process provides an atom-economical green way for the conversion of LA into GVL.

 Please wait while we load your content...
Please wait while we load your content...