Synergetic catalysis of palladium nanoparticles encaged within amine-functionalized UiO-66 in the hydrodeoxygenation of vanillin in water†
Abstract
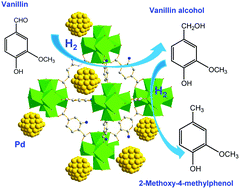
Ultrasmall palladium nanoparticles (1.5–2.5 nm) encapsulated in metal–organic frameworks (MOFs) have been prepared by introducing a palladium precursor into a highly porous and hydrothermally stable amine-functionalized UiO-66 (NH2-UiO-66) via a direct anionic exchange and subsequent H2 reduction. The prepared Pd@NH2-UiO-66 catalyst was then applied in the hydrodeoxygenation of vanillin (a typical model compound of lignin) at a low H2 pressure in aqueous media. Excellent catalytic results (100% conversion of vanillin with exclusive selectivity for 2-methoxy-4-methylphenol) could be achieved over the developed 2.0 wt% Pd@NH2-UiO-66 catalyst under mild conditions. Furthermore, the catalytic activity and selectivity were not affected after six reaction cycles indicating excellent stability and reproducibility of this catalyst system. It was found that the presence of free amine groups in the framework of NH2-UiO-66 plays a key role in the formation of uniform, well-dispersed and leaching resistant palladium nanoparticles within the MOF host. Moreover, the developed Pd@NH2-UiO-66 exhibits a novel synergetic catalysis in the hydrodeoxygenation due to the cooperation between the well-dispersed metallic Pd sites and the amine-functionalized MOF support, in which Pd offers hydrogenation activity and the MOF support facilitates hydrogenolysis of the intermediate vanillin alcohol to the 2-methoxy-4-methylphenol product.

 Please wait while we load your content...
Please wait while we load your content...