Sodium ion interaction with psyllium husk (Plantago sp.)†
Abstract
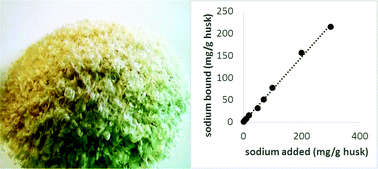
The nature of and factors effecting sodium interactions with psyllium were investigated in vitro. In a batch extraction system, psyllium mucilage gel retained at least 50% of sodium across a range of concentrations (5–300 mg sodium per g psyllium) and pH (2–10) environments. FTIR and Na NMR analyses of psyllium gels indicated that binding was complex with non-specific multi-site interactions. The potential use of psyllium husk as a binding agent for the reduction of bioavailable sodium was therefore evaluated. The binding of sodium at physiologically relevant conditions (pH 1.2 (stomach) and 6.8 (intestine)) was studied in a gastrointestinal tract (GIT) pH simulated model. Results show consistently high sodium retention (∼50%) across the GIT model and less than 20% loss of bound sodium under the simulated intestinal pH conditions after repeated washings.


 Please wait while we load your content...
Please wait while we load your content...