Modelling of the production of ACE inhibitory hydrolysates of horse mackerel using proteases mixtures
Abstract
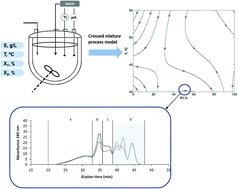
Fish protein hydrolysates from Mediterranean horse mackerel were produced by using a mixture of two commercial endoproteases (i.e. subtilisin and trypsin) at different levels of substrate concentration (2.5 g L−1, 5 g L−1, and 7.5 g L−1 of protein), temperature (40 °C, 47.5 °C, and 55 °C) and percentage of subtilisin in the enzyme mixture (0%, 25%, 50%, 75% and 100%). A crossed mixture process model was employed to predict the degree of hydrolysis (DH) and the ACE inhibitory activity of the final hydrolysates as a function of the experimental factors. Both models were optimized for a maximum DH and ACE inhibition. A maximum DH (17.1%) was predicted at 2.54 g L−1 of substrate concentration, 40 °C and an enzyme mixture comprising 38.3% of subtilisin and 61.7% of trypsin. Although its proteolytic activity is limited, the presence of trypsin in the enzyme mixture allowed obtaining higher degrees of hydrolysis at low temperatures, which is desirable to minimize thermal deactivation of the proteins. Similarly, a percentage of ACE inhibition above 48% was attained at 2.5 g L−1 of protein, 40 °C and a 1 : 1 mixture of both proteases. Higher values of ACE inhibition could be attained by increasing both the temperature and the amount of trypsin in the enzyme mixture (e.g. 50% ACE inhibition at 55 °C and 81.5% of trypsin). Finally, those hydrolysates exhibiting the highest levels of ACE inhibition were subjected to simulated gastrointestinal digestion. These assays confirmed the resistance of active fractions against their degradation by digestive enzymes.

 Please wait while we load your content...
Please wait while we load your content...