Relevant pH and lipase for in vitro models of gastric digestion†
Abstract
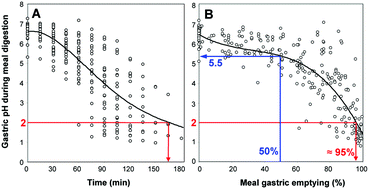
The development of in vitro digestion models relies on the availability of in vivo data such as digestive enzyme levels and pH values recorded in the course of meal digestion. The variations of these parameters along the GI tract are important for designing dynamic digestion models but also static models for which the choice of representative conditions of the gastric and intestinal conditions is critical. Simulating gastric digestion with a static model and a single set of parameters is particularly challenging because the variations in pH and enzyme concentration occurring in the stomach are much broader than those occurring in the small intestine. A review of the literature on this topic reveals that most models of gastric digestion use very low pH values that are not representative of the fed conditions. This is illustrated here by showing the variations in gastric pH as a function of meal gastric emptying instead of time. This representation highlights those pH values that are the most relevant for testing meal digestion in the stomach. Gastric lipolysis is still largely ignored or is performed with microbial lipases. In vivo data on gastric lipase and lipolysis have however been collected in humans and dogs during test meals. The biochemical characterization of gastric lipase has shown that this enzyme is rather unique among lipases: (i) stability and activity in the pH range 2 to 7 with an optimum at pH 4–5.4; (ii) high tensioactivity that allows resistance to bile salts and penetration into phospholipid layers covering TAG droplets; (iii) sn-3 stereospecificity for TAG hydrolysis; and (iv) resistance to pepsin. Most of these properties have been known for more than two decades and should provide a rational basis for the replacement of gastric lipase by other lipases when gastric lipase is not available.

 Please wait while we load your content...
Please wait while we load your content...