Photochemical transformations of thiolated polyethylene glycol coatings on gold nanoparticles†
Abstract
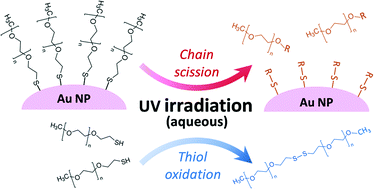
Photochemical reactions can cause significant transformations of manufactured nanomaterials in sunlit environments. While transformations of inorganic nanoparticles (NPs) have been investigated extensively, less attention has been focused on the direct impact of aqueous photochemical reactions on adsorbed organic macromolecules that form the NP corona and strongly influence the surface interactions and reactivity that affect NP transport, fate, and toxicity. Here, we assess the transformations of methoxy polyethylene glycol thiol (mPEGSH) coatings on gold NPs (AuNPs) under controlled UV irradiation. A decrease in the adsorbed layer thickness of polymer was observed within 24 h of UV irradiation, resulting in increased susceptibility of the transformed NPs to aggregation. Surface chemistry analyses, including X-ray photoelectron spectroscopy (XPS), showed loss of the ether groups but persistence of reduced S on the AuNP surface, indicative of a chain scission mechanism yielding different NP surface properties from that of either the initial PEGylated AuNP or the citrate-stabilized AuNP prior to coating with mPEGSH. The transformation of the chemisorbed polymer was compared to that of dissolved mPEGSH in the presence and absence of the Au NPs, as evaluated by liquid chromatography-mass spectrometry (LC-MS). In contrast to the NP-adsorbed coating, the primary observed transformation of the dissolved mPEGSH was thiol oxidation to disulfides without extensive chain scission. This study demonstrates that transformations of adsorbed macromolecular coatings must be considered to accurately predict NP attachment behavior, and hence transport, in environmental systems. Because the corona transformation was not predictable from that of the dissolved polymer, direct NP surface characterization is required to discern the fundamental reactions involved in the photochemical transformation of coatings after sorption to the NP surface.


 Please wait while we load your content...
Please wait while we load your content...