Aggregation and interactions of chemical mechanical planarization nanoparticles with model biological membranes: role of phosphate adsorption†
Abstract
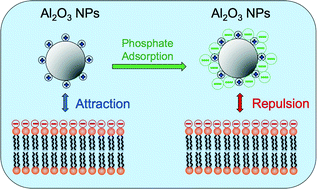
In the semiconductor industry, chemical mechanical planarization (CMP) slurries are used in large quantities for the planarization of wafers and the release of CMP nanoparticles (NPs) into the natural environment may pose a threat to human health. In this study, the aggregation behavior of four model CMP NPs, namely, colloidal SiO2, fumed SiO2, CeO2, and Al2O3 NPs, was investigated through dynamic light scattering in the presence and absence of phosphate at pH 7.4. The colloidal and fumed SiO2 NPs were observed to have remarkable stability under neutral pH conditions both in the presence and absence of phosphate. Phosphate adsorption enhanced the negative surface charge of CeO2 NPs and reversed the charge of Al2O3 NPs, resulting in an increase in the stability of both NPs. In order to evaluate the propensity for the CMP NPs to attach to and disrupt cell membranes, the interactions of these NPs with supported lipid bilayers (SLBs) and lipid vesicles composed of zwitterionic 1,2-dioleoyl-sn-glycero-3-phosphocholine (DOPC) were examined using a quartz crystal microbalance. The attachment efficiencies of both SiO2 NPs on SLBs increased to 1 when the NaCl concentration was raised to 100 mM in the presence of phosphate. In contrast, favorable attachment of CeO2 and Al2O3 NPs to SLBs was not achieved in the presence of phosphate even at elevated NaCl concentrations, likely due to the enhancement of nanoparticle surface charge resulting from phosphate adsorption. When the supported vesicles were exposed to the SiO2 NPs, no disruption of vesicles was detected despite favorable conditions for nanoparticle–membrane association.
- This article is part of the themed collection: Sustainable Nanotechnology Organization 2014

 Please wait while we load your content...
Please wait while we load your content...