Bismuth oxide: a versatile high-capacity electrode material for rechargeable aqueous metal-ion batteries†
Abstract
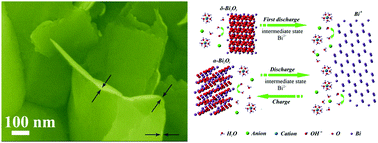
Rechargeable aqueous metal-ion (such as Li+, Na+, Mg2+, Al3+) batteries are of great importance to enrich safer, cheaper and sustainable electrochemical energy storage technologies. One of the major challenges in developing such batteries is that few electrode material systems are available to achieve prominent, reversible and stable redox reactions in aqueous electrolytes. Here we systematically report that a versatile Bi2O3 electrode material is able to electrochemically store charges in more than ten types of aqueous monovalent, divalent and trivalent metal ion electrolytes. A remarkably high specific capacity (∼357 mA h g−1 at 0.72C), outstanding rate capability (217C; 75 000 mA g−1) and good cycle life (>200 cycles) are demonstrated in a neutral mixed Li+ electrolyte. A unique “quasi-conversion reaction” charge storage mechanism that differs from a conventional intercalation-type mechanism is further unveiled (Bi2O3 ↔ Bi0). By pairing with a Li+ intercalation electrode, an aqueous LiMn2O4//Bi2O3 full cell is fabricated, which exhibits stable cycling with a low self-discharge rate and delivers a high energy density of ∼78 W h kg−1, far superior to typical aqueous lithium ion batteries (≤50 W h kg−1). Moreover, even with a relatively high mass loading of 5 mg cm−2 by slurry casting, the Bi2O3 electrode still manifests excellent performance. We anticipate that our work will stimulate the development of diverse electrode materials for aqueous rechargeable batteries.
- This article is part of the themed collection: Energy & Environmental Science Readers’ Choice Lectureship

 Please wait while we load your content...
Please wait while we load your content...