Novel layered solid oxide fuel cells with multiple-twinned Ni0.8Co0.2 nanoparticles: the key to thermally independent CO2 utilization and power-chemical cogeneration†
Abstract
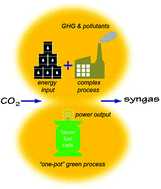
To energy-efficiently offset our carbon footprint, we herein developed a novel CH4–CO2 dry reforming process to co-produce electricity and CO-concentrated syngas, which takes advantage of the selective oxidation of H2 in high performance proton-conducting solid oxide fuel cells (SOFCs). In these cells, an additional functional layer, consisting of a Ni0.8Co0.2–La0.2Ce0.8O1.9 (NiCo–LDC) composite, was successfully incorporated into the anode support, forming a layered SOFC configuration. The multiple-twinned bimetallic nanoparticles were then proven to have superior activity towards in situ dry reforming. In comparison to the conventional design, this layered SOFC demonstrated drastically improved CO2 resistance as well as internal reforming efficiency (CO2 conversion reached 91.5% at 700 °C), and up to 100 h galvanostatic stability in a CH4–CO2 feedstream at 1 A cm−2. More importantly, H2 was effectively and exclusively converted by electrochemical oxidation, yielding no CO2 but CO concentrated syngas in the anode effluent. The maximum power density exceeded 910 mW cm−2 at 700 °C with a polarization resistance as low as 0.121 Ω cm2. Consequently, the heat released by H2 electrochemical oxidation fully compensated for that required by the extremely endothermic dry reforming reaction, making the entire process thermally self-sufficient. We also showed that the layered design was beneficial in terms of decreasing coking and increasing CO2 resistance of the SOFC in the mixed CO2 and CH4 feedstock. This novel process promises to play a pivotal role in future CO2 conversion and utilization.

 Please wait while we load your content...
Please wait while we load your content...