Bio-inspired noble metal-free nanomaterials approaching platinum performances for H2 evolution and uptake†
Abstract
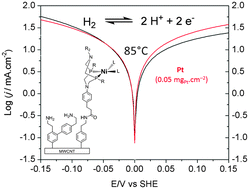
Hydrogen/water interconversion is a key reaction in the context of new energy technologies, including hydrogen fuel cells, water electrolyzers, and water-splitting photoelectrochemical cells. Specifications differ for these technologies to meet economic viability but state-of-the-art prototypes all rely on the powerful catalytic properties of the platinum metal as a catalyst for hydrogen production and uptake. Yet, this scarce and expensive metal is not itself a sustainable resource and its replacement by low cost and readily available materials is a requisite for these technologies to become economically viable. Here we revisit the preparation of bioinspired nanomaterials for hydrogen evolution and uptake (Le Goff et al., Science, 2009, 326, 1384–1387) and show that molecular engineering combined with three dimensional structuring of the electrode material allows the preparation of stable materials based on nickel bisdiphosphine catalytic units with performances in a 0.5 M sulphuric acid aqueous electrolyte that approach those of commercial platinum-based materials (0.05 mgPt cm−2) assessed under similar, technologically relevant, operational conditions.

 Please wait while we load your content...
Please wait while we load your content...