Activation of aldehydes by exocyclic iridium(i)-η4:π2-diene complexes derived from 1,3-oxazolidin-2-ones†
Abstract
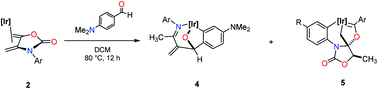
The Ir(I) complexes [TpMe2Ir(η4-1,4-diene)] 2b and 2c react thermally with a variety of aromatic aldehydes, 3a–e, to generate the metallabicyclic compounds 4e–k and the Fischer-type carbenes 5a–b in moderate yields. These reactions are proposed to take place with the initial formation of η1-aldehyde adducts as key intermediates. The formation of the metallabicyclic compounds 4e–k involves a formal decarboxylation process at the exo-2-oxazolidinone diene and an ortho metallation of the aromatic ring. The generation of the Fischer-type carbenes 5a–b is the result of a series of metal-based rearranged intermediates with no decarboxylation observed. Treatment of the η4-diene complex 2b with a variety of Lewis bases induces a change in the binding mode of the diene ligand from η4:π2 to η2:σ2 to form the Ir(III) derivatives 6b–d of composition TpMe2Ir-(η4:π2-1,4-diene)(L) (L = CO, MeCN, and C5H5N). A study of reactions of complex 2b with either mono- or poly-deuterated aldehydes was performed to understand the mechanisms of such processes. The results of these studies were used to determine plausible formation mechanisms of the metallabicyclic compounds 4e–4k and Fischer-type carbenes 5a–b compound series. These mechanisms were corroborated by density functional theory (DFT) calculations of the free energy profiles.

 Please wait while we load your content...
Please wait while we load your content...