Ligand coordination modulates reductive elimination from aluminium(iii)†
Abstract
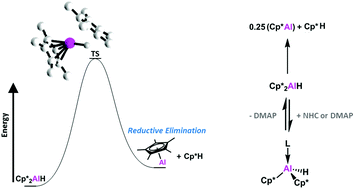
Oxidative addition of inert bonds at low-valent main-group centres is becoming a major class of reactivity for these species. The reverse reaction, reductive elimination, is possible in some cases but far rarer. Here, we present a mechanistic study of reductive elimination from Al(III) centres and unravel ligand effects in this process. Experimentally determined activation and thermodynamic parameters for the reductive elimination of Cp*H from Cp*2AlH are reported, and this reaction is found to be inhibited by the addition of Lewis bases. We find that C–H oxidative addition at Al(I) centres proceeds by initial protonation at the low-valent centre.


 Please wait while we load your content...
Please wait while we load your content...