Multi-electron redox processes at a Zr(iv) center facilitated by an appended redox-active cobalt-containing metalloligand†
Abstract
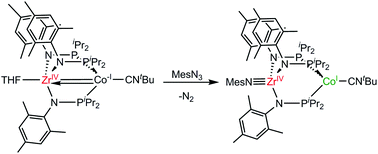
The reactivity of a reduced heterobimetallic Co−I/ZrIV complex, (tBuNC)Co(iPr2PNMes)3Zr(THF) (2), with a series of azido and diazo reagents is explored to demonstrate the feasibility of facilitating two-electron redox processes at a formally d0 Zr(IV) center using the appended Co fragment exclusively as an electron-reservoir. Addition of mesityl or adamantyl azide to 2 affords the terminal (tBuNC)Co(iPr2PNMes)3Zr![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) NMes (3) and bridging (tBuNC)Co(iPr2PNMes)2(μ-NAd)Zr(iPr2PNMes) (4) CoI/ZrIV imido products, respectively. Similarly, diphenyldiazomethane reacts with 2 to afford the terminal Ph2CN22−-bound product (tBuNC)Co(iPr2PNMes)3Zr
NMes (3) and bridging (tBuNC)Co(iPr2PNMes)2(μ-NAd)Zr(iPr2PNMes) (4) CoI/ZrIV imido products, respectively. Similarly, diphenyldiazomethane reacts with 2 to afford the terminal Ph2CN22−-bound product (tBuNC)Co(iPr2PNMes)3Zr![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N–N
N–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CPh2 (5) via a two-electron oxidation of the Co center. Thermolysis of 5 results in a structural rearrangement to the diazomethane-bridged isomer (tBuNC)Co(iPr2PNMes)2(μ-N2CPh2)Zr(iPr2PNMes) (6). In contrast, treatment of 2 with 0.5 equivalents of the conjugated diazo reagent ethyl diazoacetate affords a tetranuclear ZrIV/Co0 complex, (tBuNC)Co(iPr2PNMes)3Zr(μ2–κ1-O-η2-N,N-OC(OEt)CHN2)Zr(MesNPiPr2)3Co(CNtBu) (7), bridged through enolate and η2-bound diazo functionalities.
CPh2 (5) via a two-electron oxidation of the Co center. Thermolysis of 5 results in a structural rearrangement to the diazomethane-bridged isomer (tBuNC)Co(iPr2PNMes)2(μ-N2CPh2)Zr(iPr2PNMes) (6). In contrast, treatment of 2 with 0.5 equivalents of the conjugated diazo reagent ethyl diazoacetate affords a tetranuclear ZrIV/Co0 complex, (tBuNC)Co(iPr2PNMes)3Zr(μ2–κ1-O-η2-N,N-OC(OEt)CHN2)Zr(MesNPiPr2)3Co(CNtBu) (7), bridged through enolate and η2-bound diazo functionalities.
- This article is part of the themed collection: Recipients of the Dalton Transactions UC Berkeley Lecture

 Please wait while we load your content...
Please wait while we load your content...