The photochemistry of mono- and dinuclear cyclometalated bis(tridentate)ruthenium(ii) complexes: dual excited state deactivation and dual emission†
Abstract
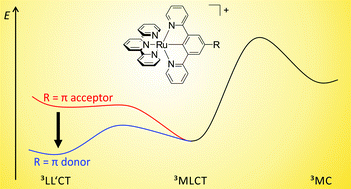
The synthesis and characterization of a series of weakly emissive mononuclear cyclometalated [Ru(dpb-R)(tpy)]+ complexes with functional groups R of varying electron-donating characters at the dpb ligand are described (dpbH = 1,3-di(2-pyridyl)benzene, tpy = 2,2′;6′,2′′-terpyridine, 1+: R = NHCOMe, 2+: R = NH2, 3+: R = COOEt, 4+: R = COOH). Steady-state emission spectroscopy in the temperature range between 298 K and 77 K revealed a previously unrecognized excited state deactivation pathway via low-lying triplet ligand-to-ligand (3LL′CT) charge transfer states in addition to the well-known pathway via3MC states. Thermal activation barriers for depopulation of the emissive metal-to-ligand charge transfer (3MLCT) states via the 3MC (metal-centered) and 3LL′CT states were determined experimentally for complexes 1+ and 3+. The experimental results were further corroborated by calculating the respective 3MLCT–3LL′CT and 3MLCT–3MC transition states and their energies with density functional theoretical methods. The R substituent modifies the energy difference between the 3MLCT and 3LL′CT states and the corresponding activation barrier but leaves the analogous 3MLCT/3MC energetics essentially untouched. Additionally, the dinuclear complex [(tpy)Ru(dpb-NHCO-dpb)Ru(tpy)]2+, 62+, containing a biscyclometalating bridge was devised. Despite the asymmetric nature induced by the amide bridge, the mixed-valent cation 63+ is ascribed to Robin–Day class II with a broad and intense intervalence charge-transfer (IVCT) absorption (λmax = 1165 nm). Upon optical excitation, the RuII/RuII complex 62+ exhibits dual emission in liquid solution from two independently emitting 3MLCT states localized at the two remote [Ru(tpy)] fragments. No equilibration via Dexter energy transfer is possible due to their large distance and short excited state lifetimes.

 Please wait while we load your content...
Please wait while we load your content...