U(v) in metal uranates: a combined experimental and theoretical study of MgUO4, CrUO4, and FeUO4†
Abstract
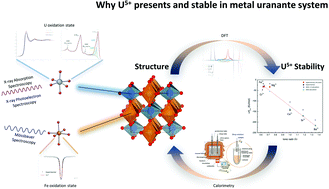
Although pentavalent uranium can exist in aqueous solution, its presence in the solid state is uncommon. Metal monouranates, MgUO4, CrUO4 and FeUO4 were synthesized for detailed structural and energetic investigations. Structural characteristics of these uranates used powder X-ray diffraction, synchrotron X-ray absorption spectroscopy, X-ray photoelectron spectroscopy, and 57Fe-Mössbauer spectroscopy. Enthalpies of formation were measured by high temperature oxide melt solution calorimetry. Density functional theory (DFT) calculations provided both structural and energetic information. The measured structural and thermodynamic properties show good consistency with those predicted from DFT. The presence of U5+ has been solidly confirmed in CrUO4 and FeUO4, which are thermodynamically stable compounds, and the origin and stability of U5+ in the system was elaborated by DFT. The structural and thermodynamic behaviour of U5+ elucidated in this work is relevant to fundamental actinide redox chemistry and to applications in the nuclear industry and radioactive waste disposal.


 Please wait while we load your content...
Please wait while we load your content...