Water based scale-up of CPO-27 synthesis for nitric oxide delivery†
Abstract
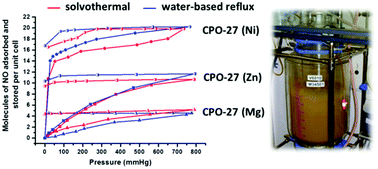
The applicability of water-based reflux and room temperature synthesis processes for the production of CPO-27 MOFs, suitable for NO delivery applications, is investigated. NO adsorption, storage and release performance of products obtained under reflux conditions are comparable to those of equivalent samples synthesised from traditional solvothermal methods at small scale. Products obtained from room temperature processes show lower NO release capability, although the quantities that are released are still more than adequate for biomedical applications. Results also reveal differences for the first time in NO uptake, storage and release depending on whether Zn, Ni or Mg is employed. The results indicate that while the crystallinity of CPO-27(Zn) and CPO-27(Mg) is not affected by moving to lower temperature methods, the crystallinity of CPO-27(Ni) is reduced. Particle morphology and size is also affected. The low temperature processes are successfully demonstrated at 20 L and 100 L scale and the main problems encountered during scale-up are outlined. The 100 L scale is in itself an appropriate production scale for some niche biomedical products. Indeed, results indicate that this synthesis approach is suitable for commercial production of MOFs for this application field. We also confirm that BET surface area from nitrogen adsorption at 77 K is not a good indicator for successful adsorption of NO.

 Please wait while we load your content...
Please wait while we load your content...