A comparison of the amorphization of zeolitic imidazolate frameworks (ZIFs) and aluminosilicate zeolites by ball-milling†
Abstract
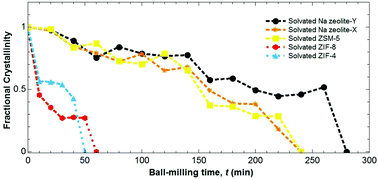
X-ray diffraction has been used to investigate the kinetics of amorphization through ball-milling at 20 Hz, for five zeolitic imidazolate frameworks (ZIFs) – ZIF-8, ZIF-4, ZIF-zni, BIF-1-Li and CdIF-1. We find that the rates of amorphization for the zinc-containing ZIFs increase with increasing solvent accessible volume (SAV) in the sequence ZIF-8 > ZIF-4 > ZIF-zni. The Li–B analogue of the dense ZIF-zni amorphizes more slowly than the corresponding zinc phase, with the behaviour showing a correlation with their relative bulk moduli and SAVs. The cadmium analogue of ZIF-8 (CdIF-1) amorphizes more rapidly than the zinc counterpart, which we ascribe primarily to its relatively weak M–N bonds as well as the higher SAV. The results for the ZIFs are compared to three classical zeolites – Na-X, Na-Y and ZSM-5 – with these taking up to four times longer to amorphize. The presence of adsorbed solvent in the pores is found to render both ZIF and zeolite frameworks more resistant to amorphization. X-ray total scattering measurements show that amorphous ZIF-zni is structurally indistinguishable from amorphous ZIF-4 with both structures retaining the same short-range order that is present in their crystalline precursors. By contrast, both X-ray total scattering measurements and 113Cd NMR measurements point to changes in the local environment of amorphous CdIF-1 compared with its crystalline CdIF-1 precursor.
- This article is part of the themed collections: In celebration of Tony Cheetham’s 70th birthday and Flexibility and Disorder in Metal-Organic Frameworks


 Please wait while we load your content...
Please wait while we load your content...