Solubilisation and chemical fixation of copper(ii) in micronized copper treated wood
Abstract
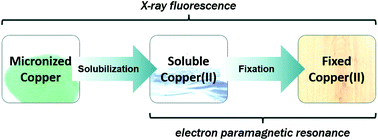
Wood preservatives using micronized particulate copper as the main active ingredient recently introduced in the USA have generated controversies due to their limited intrinsic solubility compared to the conventional soluble copper treatments. Because the availability of soluble copper ions is essential for these preservatives to provide an effective treatment, concerns centered on whether they are able to produce soluble copper when introduced into wood, and the copper fixation mechanism of the treatment is little understood. A combination of Electron Paramagnetic Resonance (EPR) spectroscopy and X-ray Fluorescence (XRF) spectroscopy has been successfully utilized to characterize and quantify solubilized and fixed copper species in micronized copper treated wood, which also contains unreacted particulate copper. On the basis of the experimental results, the fixation mechanism is thought to be triggered by the reaction between the carboxylic acid protons in hemicellulose of wood with the particulate copper, and the quantities of the solubilized and fixed copper species are determined by the availability of these acidic protons.

 Please wait while we load your content...
Please wait while we load your content...