Catalysis of solar hydrogen production by iron atoms on the surface of Fe-doped silicon carbide†
Abstract
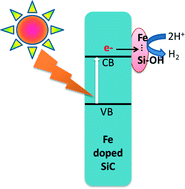
The activity of silicon carbide nanowires for light-driven hydrogen production is increased 100-fold by doping with Fe. The surface of the nanowires comprises Si, C, O (OH) and Fe atoms: importantly, selective removal of surface Fe by treatment with HF removes >99% of catalytic H2 evolution activity without altering the bulk electronic properties. Laboratory experiments with an aqueous suspension of nanoparticles at pH 6.4 reveal high stability and a quantum efficiency of 11.6% at 420 nm, when iodide is used as electron donor. The results show that Fe must be a component of surface catalytic sites that include neighbouring OH functionalities, and add further assurance that scalable artificial photosynthesis can be achieved using inexpensive and abundant resources.

 Please wait while we load your content...
Please wait while we load your content...