Single molecule study of samarium oxide nanoparticles as a purely heterogeneous catalyst for one-pot aldehyde chemistry†‡
Abstract
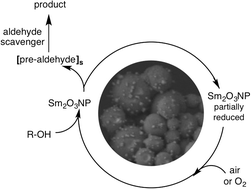
Heterogeneous catalysis holds distinct advantages over homogeneous catalysis; however, it is only truly advantageous if unaffected by metal ion leaching or in situ formation of a soluble catalytically active species. Herein, samarium oxide nanoparticles (Sm2O3NP) were employed as a redox catalyst for the first time, providing a generally applicable route to performing one-pot aldehyde chemistry beginning with an inexpensive and more readily-prepared starting material – an alcohol. The reaction is efficient under mild conditions and does not suffer from over-oxidation of the starting material to its corresponding carboxylic acid. This key asset of the Sm2O3NP catalyst hinges upon a lack of release of free aldehyde into the system. Single molecule fluorescence microscopy revealed that catalysis is restricted to the surfaces of small nanoparticles in a polydisperse nanomaterial, where the product of the oxidative process can be selectively revealed via a subsequent coupling reaction, which occurs without the intermediacy of a free aldehyde. Instead the catalytically active particles act as docking stations, holding the pre-aldehyde until intercepted by a second reagent to complete the oxidation and subsequent reaction in a single step. Coupling this supramolecular strategy to our single molecule approach revealed the interesting nature of the catalytic mechanism and demonstrated that the catalysis is a purely heterogeneous process. Samarium is quite abundant relative to many transition metals (e.g. Ru, Ir, Pt) and thus Sm2O3NP-based nanomaterials may present an opportunity to develop more sustainable catalysts for common organic transformations.

 Please wait while we load your content...
Please wait while we load your content...