Enhancement of photocatalytic H2 production activity of CdS nanorods by cobalt-based cocatalyst modification†
Abstract
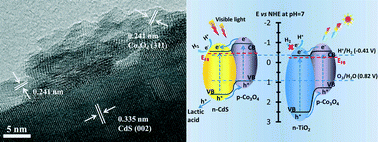
Decreasing the recombination of photogenerated charge carriers in photocatalysts is critical to enhancing the efficiency of photocatalytic H2 production by water splitting. In this paper, we report the enhancement of visible-light photocatalytic H2 production activity by introducing various earth-abundant cobalt species onto CdS nanorods to facilitate charge transport and separation. The modification of Co(OH)2, Co3O4, CoS, CoO and Co2+ as a cocatalyst enhanced the photocatalytic H2 production activity of CdS nanorods under visible light irradiation. Among them, the as-prepared CdS–Co3O4 sample exhibited the highest photocatalytic H2 production rate of 150.7 μmol h−1 with a quantum efficiency of 9.7% at 420 nm, even without Pt as a cocatalyst. This outstanding activity arises from the formation of p–n heterojunctions at the interface of the n-type CdS nanorods and p-type Co3O4 nanoparticles, which can facilitate the charge separation and transport due to the effect of utilizing the internal electric field. Electrochemical measurement results further proved that the observed superiority of the CdS–Co3O4 arises from the effect of the p–n heterojunction including enhancement of light-harvesting capacity, and improvement of charge transfer and separation. To our surprise, the loading of various Co species as a cocatalyst onto Degussa P25 TiO2 did not show an enhanced photocatalytic H2 production activity due to the unsuitable positions of the band-gap edge of Co-based TiO2 heterojunctions in photocatalytic H2 production. This work demonstrated for the first time that the low-cost Co3O4 nanocluster is the most suitable Co-based cocatalyst to replace noble metals for photocatalytic H2 production in the CdS system.

 Please wait while we load your content...
Please wait while we load your content...