High catalytic activity of mesoporous Co–N/C catalysts for aerobic oxidative synthesis of nitriles†
Abstract
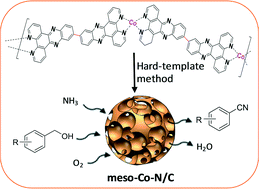
A high-efficiency and atom-economic synthetic strategy for nitriles by aerobic ammoxidation of alcohols is developed using a novel mesoporous cobalt-coordinated nitrogen-doped carbon catalyst (meso-Co–N/C) fabricated from a cobalt-coordinating polymer, which manifests superior activity towards the target reaction. The catalytic system features a broad substrate scope for various benzylic, allylic as well as heterocyclic alcohols, providing good to excellent yields of the target products with high selectivities, albeit with 0.5 mol% Co catalyst loading. 11,11′-Bis(dipyrido[3,2-a:2′,3′-c]phenazinyl) (bidppz) with extreme thermostability is selected as a robust ligand bridge between cobalt ions, resulting in the homogeneous distribution of active sites at the atomic or subnanoscale level and high catalyst yield. Silica colloid or ordered mesoporous silica SBA-15 is employed to realize the mesoporous structure. The unprecedented performance of the meso-Co–N/C catalyst is attributed to its high Brunauer–Emmett–Teller (BET) surface area (up to 680 m2 g−1) with a well-controlled mesoporous structure and homogeneous distribution of active sites. Kinetic analysis demonstrates that the catalytic oxidation of benzyl alcohol to benzaldehyde is the turnover-limiting step and that the apparent activation energy for benzonitrile synthesis is 61.5 kJ mol−1 and cationic species are involved in the reaction.

 Please wait while we load your content...
Please wait while we load your content...