Promoting effects of MgO, (NH4)2SO4 or MoO3 modification in oxidative esterification of methacrolein over Au/Ce0.6Zr0.4O2-based catalysts†
Abstract
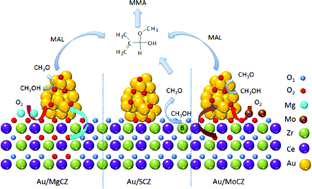
MgO, (NH4)2SO4 or MoO3-modified Ce0.6Zr0.4O2-supported gold catalysts were synthesized and studied for the oxidative esterification of methacrolein with methanol to methyl methacrylate. Although the particle size distribution and oxidative state of the supported Au showed almost no change before and after Ce0.6Zr0.4O2 modification as confirmed by TEM, Raman and XPS technologies, all of the modified Au/Ce0.6Zr0.4O2 catalysts demonstrated highly improved catalytic activities. H2-TPR patterns indicated that the amounts of low-temperature active oxygen species increased in the MgO- and MoO3-modified catalysts. At the same time, the strong interaction between gold and Ce–Zr oxides in the MgO-modified catalyst and the charge transfer between Mo6+ and Ce3+ in the MoO3-modified catalyst were evidenced by XPS characterization. Based on the enhanced synergetic effects between the Au nanoparticles and the support for oxygen activation, the MgO-promoted Au catalyst presented the best performance with a methacrolein conversion of 98% and a methyl methacrylate selectivity of 90% in the absence of a base at 80 °C for 2 h of reaction. Differing from MgO and MoO3, (NH4)2SO4 modification simultaneously increased the acid–base properties of the catalyst and then increased the activity of the catalyst due to the promoted formation of the intermediate hemiacetal.

 Please wait while we load your content...
Please wait while we load your content...