Improving the catalytic performance of SAPO-18 for the methanol-to-olefins (MTO) reaction by controlling the Si distribution and crystal size†
Abstract
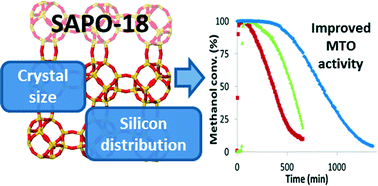
The physico-chemical properties of the small pore SAPO-18 zeotype have been controlled by properly selecting the organic molecules acting as organic structure directing agents (OSDAs). The two organic molecules selected to attempt the synthesis of the SAPO-18 materials were N,N-diisopropylethylamine (DIPEA) and N,N-dimethyl-3,5-dimethylpiperidinium (DMDMP). On the one hand, DIPEA allows small crystal sizes (0.1–0.3 μm) to be attained with limited silicon distributions when the silicon content in the synthesis gel is high (Si/TO2 ∼0.8). On the other hand, the use of DMDMP directs the formation of larger crystallites (0.9–1.0 μm) with excellent silicon distributions, even when the silicon content in the synthesis media is high (Si/TO2 ∼0.8). It is worth noting that this is the first description of the use of DMDMP as OSDA for the synthesis of the SAPO-18 material, revealing not only the excellent directing role of this OSDA in stabilizing the large cavities present in the SAPO-18 structure, but also its role in selectively placing the silicon atoms in isolated framework positions. The synthesized SAPO-18 materials have been characterized by different techniques, such as powder X-ray diffraction (PXRD), scanning electron microscopy (SEM), N2 adsorption, solid state NMR, and ammonia temperature programmed desorption (NH3-TPD). Finally, their catalytic activity has been evaluated for the methanol-to-olefin (MTO) process at different reaction temperatures (350 and 400 °C), revealing that the SAPO-18 catalysts with optimized silicon distributions and crystal sizes show excellent catalytic properties for the MTO reaction. These optimized SAPO-18 materials present improved catalyst lifetimes compared to standard SAPO-34 and SSZ-39 catalysts, even when tested at low reaction temperatures (i.e. 350 °C).
- This article is part of the themed collection: Catalysis on Zeolites

 Please wait while we load your content...
Please wait while we load your content...