Ag3PO4 immobilized on hydroxy-metal pillared montmorillonite for the visible light driven degradation of acid red 18†
Abstract
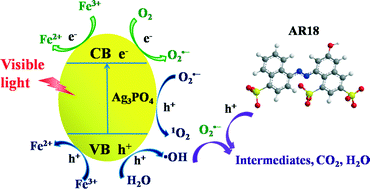
This work reports the facile fabrication of Ag3PO4/Fe–Al/Mt and Ag3PO4/Al/Mt by loading Ag3PO4 on hydroxy-iron–aluminum pillared montmorillonite (Fe–Al/Mt) and hydroxy-aluminum pillared montmorillonite (Al/Mt). The structural characteristics of the resulting materials were studied with XRD, SEM-EDS, XPS, ICP, nitrogen adsorption–desorption isotherms, and UV-vis diffuse reflectance spectra; the photocatalytic activity of the obtained catalysts was tested using acid red 18 (AR18) as a model contaminant under visible light irradiation. The obtained results illustrate that Ag3PO4 of a high dispersity and smaller size was successfully loaded on hydroxy-metal pillared montmorillonite. The photocatalytic activity and structural stability of the three synthesized catalysts were in the order Ag3PO4/Fe–Al/Mt > Ag3PO4/Al/Mt > Ag3PO4. An efficiency of 98.5% was achieved for AR18 degradation by Ag3PO4/Fe–Al/Mt after recycling seven times, while only 54.9% was achieved for Ag3PO4. The superoxide radical anion (O2˙−) was confirmed to be the dominant reactive species in all the three degradation systems, and the Ag3PO4/Fe–Al/Mt system formed the largest amount of O2˙−. Except for the larger specific surface area and smaller particle size, the high removal efficiency of AR18, remarkable O2˙− generation performance, and good stability of Ag3PO4/Fe–Al/Mt could be attributed to the presence of Fe3+ as well, which can act as an electron acceptor for photo-induced electrons from Ag3PO4 during the photocatalytic process and then inhibit the transformation of Ag+ into metallic Ag.

 Please wait while we load your content...
Please wait while we load your content...