Bidimensional ZSM-5 zeolites probed as catalysts for polyethylene cracking†
Abstract
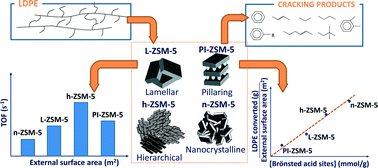
Lamellar and pillared ZSM-5 zeolites (L-ZSM-5 and PI-ZSM-5, respectively) were synthesized and tested in the catalytic cracking of low-density polyethylene (LDPE). The introduction of silica pillars into lamellar ZSM-5 caused a high increase in the Si/Al ratio (from 33 up to 64) and the generation of uniform mesopores with a size of about 3.5 nm. Both samples provided quite similar LDPE conversions at the three reaction temperatures investigated (340, 360 and 380 °C) despite the lower concentration of acid sites in PI-ZSM-5, which is assigned to the improved active centre accessibility due to the pillaring treatment. Significant activity was observed even at the lowest temperature, with LDPE conversions in the range 27–36%, which indicates that 2D ZSM-5 zeolites are convenient catalysts for polyethylene cracking. The main products of LDPE catalytic cracking were C2–C5 olefins with a selectivity of 60–70%, denoting that an end-chain cracking mechanism is predominant. 2D ZSM-5 samples were subsequently compared with nanocrystalline (n-ZSM-5) and hierarchical ZSM-5 (h-ZSM-5) zeolites. Pyridine adsorption followed by FTIR measurements showed significant differences in terms of not only acid site concentration but also the Brønsted/Lewis acid distribution among the samples. When the LDPE cracking conversion was referred to the zeolite mesopore/external surface area, a good correlation was observed with the concentration of Brønsted acid sites but not when considering just the Lewis acid sites. This interesting fact suggests that Brønsted acid sites are mainly the active centers for the cracking of the LDPE chains, concluding that in addition to the accessibility, the nature of the acid sites plays a major role in this type of reaction.
- This article is part of the themed collection: Catalysis on Zeolites

 Please wait while we load your content...
Please wait while we load your content...