One-pot two-step rapid synthesis of 3-aminopropyltrimethoxysilane-mediated highly catalytic Ag@(PdAu) trimetallic nanoparticles†
Abstract
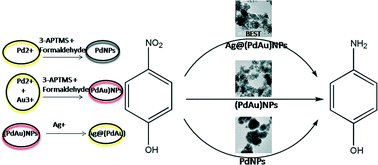
Synergistic interactions between individual components of multimetallic nanoparticles result in dramatic changes in important physical or chemical properties that outclass those of monometallic nanoparticles in many aspects. Rapid synthesis under ambient conditions of multimetallic nanoparticles having potential for conversion from a homogeneous suspension to a solid matrix without compromising the catalyzing ability remains a daunting challenge. In an attempt to meet such challenges, trimetallic Ag@(PdAu) nanoparticles involving the active role of 3-aminopropyltrimethoxysilane (3-APTMS) were fabricated and evaluated as catalysts in both homogeneous and heterogeneous phases using the model reaction of p-nitrophenol reduction [k = 0.14 ± 0.0025 s−1 for Ag@(PdAu)NPs]. The synthesis process involves the sequential addition of Ag+ over (PdAu) bimetallic nanoparticles, made by simultaneous reduction of 3-APTMS-capped metal cations (Pd2+, Au3+) in the presence of formaldehyde. 3-APTMS not only assists in nanoparticle synthesis but also promotes the conversion of a homogeneous nanoparticle suspension to a solid matrix, a prerequisite for heterogeneous catalysts, by initiating Si–O–Si linkages in the presence of hydrochloric acid. The results based on UV-vis spectroscopy, transmission electron microscopy and X-ray photoelectron spectroscopy clearly demonstrate the synthesis of trimetallic nanoparticles within seconds under ambient conditions. Characterization data reveal that AgNPs not only form alloys with AuNPs but also form pear-shaped structures physically attached over simultaneously made (PdAu) core–shell nanoparticles, when present in excess. A gradual increase in the catalytic behavior of the nanomaterials is recorded in the order of PdNPs < (PdAu)NPs < Ag@(PdAu)NPs.

 Please wait while we load your content...
Please wait while we load your content...