How TiO2 facets determine arsenic adsorption and photooxidation: spectroscopic and DFT studies†
Abstract
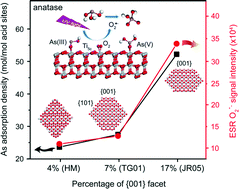
Anatase TiO2 nanomaterials have been widely used in arsenic (As) remediation, although reports on their adsorption and photocatalytic capacity have been controversial. The motivation for our study is to explore the As adsorption and photooxidation processes on different TiO2 facets at the molecular level. Our results from multiple complementary characterization techniques suggest that anatase {001} facets have stronger Lewis acid sites than those on {101} facets, resulting in a higher As adsorption affinity. Density functional theory (DFT) calculations confirmed that the As surface complex is more energetically favorable on {001} than on the {101} facets. In addition, the strong interaction of the {001} facets with molecular O2 facilitates the transfer of photo-excited electrons to the adsorbed O2 to generate a superoxide radical (O2˙−), which is the primary As(III) oxidant as evidenced by our radical-trapping experiments. Meanwhile, the oxygen vacancies on the {001} facets could expedite the interfacial electron transfer and electron–hole separation, which promote the generation of O2˙− and, ultimately, the catalytic efficiency. The insights gained from this study provide a firm basis for the proposition that As adsorption and photoactivity can be mediated by tailoring the exposed TiO2 facets, which is of essence in the design and application of TiO2-based environmental technologies.

 Please wait while we load your content...
Please wait while we load your content...