Catalytic performance of Pd-promoted Cu hydrotalcite-derived catalysts in partial hydrogenation of acetylene: effect of Pd–Cu alloy formation†
Abstract
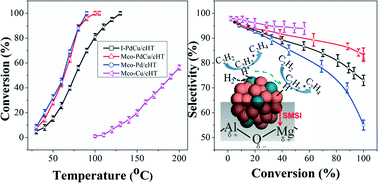
Aiming at the preparation of catalysts with well-controlled structures, bimetallic PdCu catalysts containing only small amounts of Pd were prepared by two different methods for partial hydrogenation of acetylene. The highly dispersed and stable PdCu catalyst with the Pd : Cu atomic ratio of 1 : 40 was obtained from a Pd(OH)2/CuMgAl hydrotalcite (HT) precursor by a modified co-precipitation method. The obtained PdCu catalyst possessed uniform PdCu nanoalloys with an average size of 1.8 ± 0.3 nm. As a comparison, the impregnation method was also employed to prepare bimetallic PdCu catalysts and it was confirmed that the Pd-rich(core)–Cu-rich(shell) structure was dominant. Under identical reaction conditions, approximately 100% conversion and 82% selectivity at 100 °C were achieved using the PdCu nanoalloy catalyst which were 23% and 12% higher than those of the Pd-rich(core)–Cu-rich(shell) catalyst. The preferable activity was ascribed to the homogeneous nanoalloy structure and small size effect. The enhanced selectivity was attributed to the strong synergistic effect of PdCu. More significantly, this superior catalytic performance can be retained after 48 h of continuous reaction due to the excellent resistibility against carbon deposition and the confinement effect.

 Please wait while we load your content...
Please wait while we load your content...