Deactivation of Cu-SSZ-13 by SO2 exposure under SCR conditions
Abstract
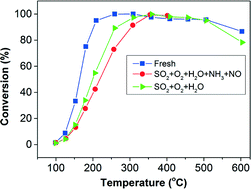
A deactivation study of Cu-SSZ-13 has been conducted using SO2 exposure under SCR conditions and examining its effect on different reactions involving NH3-SCR. Several reactions, including NH3 storage/TPD, NO/NH3 oxidation, standard SCR, fast SCR and SCR with 75% NO2, as well as NH3–NO2 storage/TPD, were investigated at a temperature range of 100–400 °C after exposing the catalyst to 30 ppm SO2 under SCR conditions at 300 °C for 90 min. The catalyst was characterized using XRD, BET, ICP-SFMS and H2-TPR. The BET surface area and pore volume decreased after the sulfur treatment presumably due to blocking by sulfur and/or ammonium–sulfur species. It was found that sulfur was not uniformly deposited along the monolith channel. The deposition occurred from the inlet towards the outlet, as evident from ICP-SFMS measurements. Part of the sulfur was removed after an SCR experiment up to 400 °C. However, this removal was observed only in the inlet half of the sample and not in the outlet. Ammonia TPD experiments revealed that the sulfur poisoning resulted in additional sites that were capable of adsorbing ammonia, resulting in increased ammonia storage. Moreover, standard SCR was significantly deactivated by SO2 poisoning under SCR conditions. Due to the site-blocking effect of the ammonium–sulfur species, fewer copper sites are likely available for the redox SCR cycle. Furthermore, the effect of sulfur poisoning on NH3 oxidation and NO2-SCR as well as N2O production in various SCR reactions were observed. Finally, it was found that the conditions for the sulfur poisoning were critical in which SO2 deactivation under SCR conditions (NH3 + NO + O2 + H2O) was more severe compared to SO2 poisoning in O2 + H2O alone.
- This article is part of the themed collection: Catalysis on Zeolites

 Please wait while we load your content...
Please wait while we load your content...