(Hetero)aromatics from dienynes, enediynes and enyne–allenes
Abstract
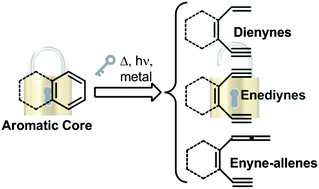
The construction of aromatic rings has become a key objective for organic chemists. While several strategies have been developed for the functionalization of pre-formed aromatic rings, the direct construction of an aromatic core starting from polyunsaturated systems is yet a less explored field. The potential of such reactions in the formation of aromatics increased at a regular pace in the last few years. Nowadays, there are reliable and well-established procedures to prepare polyenic derivatives, such as dienynes, enediynes, enyne–allenes and hetero-analogues. This has stimulated their use in the development of innovative cycloaromatizations. Different examples have recently emerged, suggesting large potential of this strategy in the preparation of (hetero)aromatics. Accordingly, this review highlights the recent advancements in this field and describes the different conditions exploited to trigger the process, including thermal and photochemical activation, as well as the use of transition metal catalysis and the addition of electrophiles/nucleophiles or radical species.


 Please wait while we load your content...
Please wait while we load your content...