Selective gas capture via kinetic trapping†
Abstract
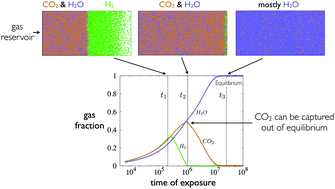
Conventional approaches to the capture of CO2 by metal–organic frameworks focus on equilibrium conditions, and frameworks that contain little CO2 in equilibrium are often rejected as carbon-capture materials. Here we use a statistical mechanical model, parameterized by quantum mechanical data, to suggest that metal–organic frameworks can be used to separate CO2 from a typical flue gas mixture when used under nonequilibrium conditions. The origin of this selectivity is an emergent gas-separation mechanism that results from the acquisition by different gas types of different mobilities within a crowded framework. The resulting distribution of gas types within the framework is in general spatially and dynamically heterogeneous. Our results suggest that relaxing the requirement of equilibrium can substantially increase the parameter space of conditions and materials for which selective gas capture can be effected.

 Please wait while we load your content...
Please wait while we load your content...