Ion mobility spectrometry-mass spectrometry examination of the structures, stabilities, and extents of hydration of dimethylamine–sulfuric acid clusters†
Abstract
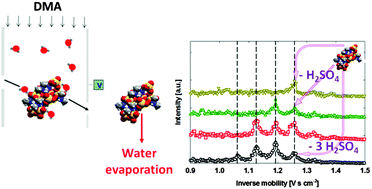
We applied an atmospheric pressure differential mobility analyzer (DMA) coupled to a time-of-flight mass spectrometer to examine the stability, mass–mobility relationship, and extent of hydration of dimethylamine–sulfuric acid cluster ions, which are of relevance to nucleation in ambient air. Cluster ions were generated by electrospray ionization and were of the form: [H((CH3)2NH)x(H2SO4)y]+ and [(HSO4)((CH3)2NH)x(H2SO4)y]−, where 4 ≤ x ≤ 8, and 5 ≤ y ≤ 12. Under dry conditions, we find that positively charged cluster ions dissociated via loss of both multiple dimethylamine and sulfuric acid molecules after mobility analysis but prior to mass analysis, and few parent ions were detected in the mass spectrometer. Dissociation also occurred for negative ions, but to a lesser extent than for positive ions for the same mass spectrometer inlet conditions. Under humidified conditions (relative humidities up to 30% in the DMA), positively charged cluster ion dissociation in the mass spectrometer inlet was mitigated and occurred primarily by H2SO4 loss from ions containing excess acid molecules. DMA measurements were used to infer collision cross sections (CCSs) for all identifiable cluster ions. Stokes–Millikan equation and diffuse/inelastic gas molecule scattering predicted CCSs overestimate measured CCSs by more than 15%, while elastic–specular collision model predictions are in good agreement with measurements. Finally, cluster ion hydration was examined by monitoring changes in CCSs with increasing relative humidity. All examined cluster ions showed a modest amount of water molecule adsorption, with percentage increases in CCS smaller than 10%. The extent of hydration correlates directly with cluster ion acidity for positive ions.


 Please wait while we load your content...
Please wait while we load your content...