Elucidating dominant pathways of the nano-particle self-assembly process†
Abstract
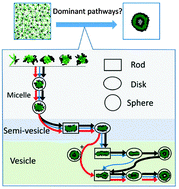
Self-assembly processes play a key role in the fabrication of functional nano-structures with widespread application in drug delivery and micro-reactors. In addition to the thermodynamics, the kinetics of the self-assembled nano-structures also play an important role in determining the formed structures. However, as the self-assembly process is often highly heterogeneous, systematic elucidation of the dominant kinetic pathways of self-assembly is challenging. Here, based on mass flow, we developed a new method for the construction of kinetic network models and applied it to identify the dominant kinetic pathways for the self-assembly of star-like block copolymers. We found that the dominant pathways are controlled by two competing kinetic parameters: the encounter time Te, characterizing the frequency of collision and the transition time Tt for the aggregate morphology change from rod to sphere. Interestingly, two distinct self-assembly mechanisms, diffusion of an individual copolymer into the aggregate core and membrane closure, both appear at different stages (with different values of Tt) of a single self-assembly process. In particular, the diffusion mechanism dominates the middle-sized semi-vesicle formation stage (with large Tt), while the membrane closure mechanism dominates the large-sized vesicle formation stage (with small Tt). Through the rational design of the hydrophibicity of the copolymer, we successfully tuned the transition time Tt and altered the dominant self-assembly pathways.

 Please wait while we load your content...
Please wait while we load your content...