“Pnicogen bonds” or “chalcogen bonds”: exploiting the effect of substitution on the formation of P⋯Se noncovalent bonds†
Abstract
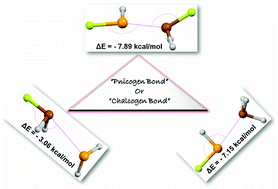
In this article, we have analyzed the nature and characteristics of P⋯Se noncovalent interactions by studying the effect of substitution on XH2P⋯SeH2, H3P⋯SeHX and XH2P⋯SeHX (X= –H, –F, –CH3, –CF3, –Cl, –OH, –OCH3, –NH2, –NHCH3, and –CN) as our systems of interest at MP2/aug-cc-pVDZ level of theory. Binding energy calculations depict that binding energy increases in the order XH2P⋯SeH2 < H3P⋯SeHX < XH2P⋯SeHX with the nature of the substituent having a direct effect on the strength of the interactions. P⋯Se contacts as short as 2.52 Å were observed and analyzed in our study. The energy values for P⋯Se contacts were found to exist in the range of −1.20 kcal mol−1 to −7.89 kcal mol−1. The topological analysis confirms the presence of P⋯Se contacts in all the complexes with characteristics similar to hydrogen bonds. NBO analysis helped in categorizing these interactions into pnicogen and chalcogen bonds, depending on the strength of P(lp) to σ*(Se–X) orbitals or Se(lp) to σ*(P–X) orbitals.

 Please wait while we load your content...
Please wait while we load your content...