Tailoring ruthenium exposure to enhance the performance of fcc platinum@ruthenium core–shell electrocatalysts in the oxygen evolution reaction†
Abstract
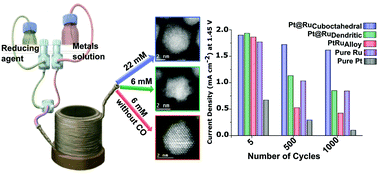
The catalytic properties of noble metal nanocrystals are a function of their size, structure, and surface composition. In particular, achieving high activity without sacrificing stability is essential for designing commercially viable catalysts. A major challenge in designing state-of-the-art Ru-based catalysts for the oxygen evolution reaction (OER), which is a key step in water splitting, is the poor stability and surface tailorability of these catalysts. In this study, we designed rapidly synthesizable size-controlled, morphology-selective, and surface-tailored platinum–ruthenium core–shell (Pt@Ru) and alloy (PtRu) nanocatalysts in a scalable continuous-flow reactor. These core–shell nanoparticles with atomically precise shells were produced in a single synthetic step with carbon monoxide as the reducing agent. By varying the metal precursor concentration, a dendritic or layer-by-layer ruthenium shell can be grown. The synthesized Pt@Ru and PtRu nanoparticles exhibit noticeably higher electrocatalytic activity in the OER compared to that of pure Pt and Ru nanoparticles. Promisingly, Pt@Ru nanocrystals with a ∼2–3 atomic layer Ru cuboctahedral shell surpass conventional Ru nanoparticles in terms of both durability and activity.

 Please wait while we load your content...
Please wait while we load your content...