Atmospheric fates of Criegee intermediates in the ozonolysis of isoprene†
Abstract
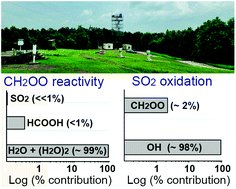
We use a large laboratory, modeling, and field dataset to investigate the isoprene + O3 reaction, with the goal of better understanding the fates of the C1 and C4 Criegee intermediates in the atmosphere. Although ozonolysis can produce several distinct Criegee intermediates, the C1 stabilized Criegee (CH2OO, 61 ± 9%) is the only one observed to react bimolecularly. We suggest that the C4 Criegees have a low stabilization fraction and propose pathways for their decomposition. Both prompt and non-prompt reactions are important in the production of OH (28% ± 5%) and formaldehyde (81% ± 16%). The yields of unimolecular products (OH, formaldehyde, methacrolein (42 ± 6%) and methyl vinyl ketone (18 ± 6%)) are fairly insensitive to water, i.e., changes in yields in response to water vapor (≤4% absolute) are within the error of the analysis. We propose a comprehensive reaction mechanism that can be incorporated into atmospheric models, which reproduces laboratory data over a wide range of relative humidities. The mechanism proposes that CH2OO + H2O (k(H2O) ∼ 1 × 10−15 cm3 molec−1 s−1) yields 73% hydroxymethyl hydroperoxide (HMHP), 6% formaldehyde + H2O2, and 21% formic acid + H2O; and CH2OO + (H2O)2 (k(H2O)2 ∼ 1 × 10−12 cm3 molec−1 s−1) yields 40% HMHP, 6% formaldehyde + H2O2, and 54% formic acid + H2O. Competitive rate determinations (kSO2/k(H2O)n=1,2 ∼ 2.2 (±0.3) × 104) and field observations suggest that water vapor is a sink for greater than 98% of CH2OO in a Southeastern US forest, even during pollution episodes ([SO2] ∼ 10 ppb). The importance of the CH2OO + (H2O)n reaction is demonstrated by high HMHP mixing ratios observed over the forest canopy. We find that CH2OO does not substantially affect the lifetime of SO2 or HCOOH in the Southeast US, e.g., CH2OO + SO2 reaction is a minor contribution (<6%) to sulfate formation. Extrapolating, these results imply that sulfate production by stabilized Criegees is likely unimportant in regions dominated by the reactivity of ozone with isoprene. In contrast, hydroperoxide, organic acid, and formaldehyde formation from isoprene ozonolysis in those areas may be significant.

 Please wait while we load your content...
Please wait while we load your content...