Predicting 17O NMR chemical shifts of polyoxometalates using density functional theory†
Abstract
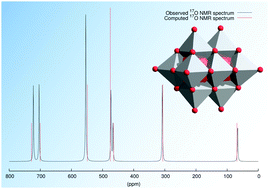
We have investigated the computation of 17O NMR chemical shifts of a wide range of polyoxometalates using density functional theory. The effects of basis sets and exchange–correlation functionals are explored, and whereas pure DFT functionals generally predict the chemical shifts of terminal oxygen sites quite well, hybrid functionals are required for the prediction of accurate chemical shifts in conjunction with linear regression. By using PBE0/def2-tzvp//PBE0/cc-pvtz(H-Ar), lanl2dz(K-) we have computed the chemical shifts of 37 polyoxometalates, corresponding to 209 17O NMR signals. We also show that at this level of theory the protonation-induced pH dependence of the chemical shift of the triprotic hexaniobate Lindqvist anion, [HxNb6O19](8−x), can be reproduced, which suggests that hypotheses regarding loci of protonation can be confidently tested.

 Please wait while we load your content...
Please wait while we load your content...