The effect of Mg2+ incorporation on the structure of calcium carbonate clusters: investigation by the anharmonic downward distortion following method
Abstract
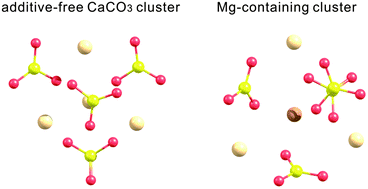
Mg2+ is considered to play an important role in the formation of calcium carbonate polymorphs; however, how it affects polymorph selection during the early stages of CaCO3 formation is not yet well understood. In the present study, in order to clarify the effect of Mg2+ on the nucleation of calcium carbonate polymorphs, the stable structures of anhydrous additive-free and Mg-containing calcium carbonate clusters are derived using the anharmonic downward distortion following method, based on quantum chemical calculations. Optimization is performed at the B3LYP/6-31+G(d) level and the solvent effect is induced by the self-consistent reaction field method using the conductor-like polarized continuum calculation model. Calculation results show that incorporating Mg2+ into clusters can change the clusters’ stable configuration. In the case of dimers and trimers, a Mg ion strongly prefers to locate at the centre of the clusters, which suggests that Mg is easy to incorporate into the clusters once it is released from its tight hydration shell. Notably, structures similar to the crystalline phase appear when only four CaCO3 units aggregate into the cluster: in the stable structure of the additive-free CaCO3 tetramer, the arrangement of Ca and CO3 ions is almost the same as that of the calcite structure, while the structure of the Mg-containing CaCO3 tetramer resembles the aragonite structure in the way that CO3 ions are stacked. These results indicate that Mg can play a key role in aragonite formation not only by inhibiting calcite growth but also by directly promoting aragonite nucleation in the early stages of CaCO3 formation.

 Please wait while we load your content...
Please wait while we load your content...