Designing a large scale synthesis strategy for high quality magnetite nanocrystals on the basis of a solution behavior regulated formation mechanism†
Abstract
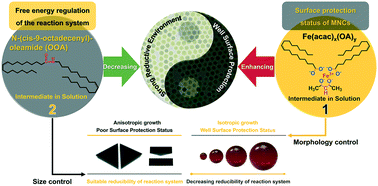
The large scale synthesis of magnetite nanocrystals (MNCs) is a sine qua non for supplying the rapidly increasing demand for MNCs in biomedical applications (e.g. magnetic resonance imaging (MRI), magnetic hyperthermia and targeted drug delivery), but it is still difficult to achieve their industrial production due to the operation complexity, strict synthetic conditions and the narrow error tolerance range of existing approaches. Here, we report a facile synthetic procedure using a solvothermal route with an iron acetylacetonate (Fe(acac)3), oleic acid (OA) and oleylamine (OAm) reaction system on a large scale, which is designed under the guidance of the solution behavior regulated formation mechanism of MNCs. Two intermediates, Fe(acac)x(OA)y and N-(cis-9-octadecenyl)-oleamide (OOA), are found to play key roles in adjusting the surface protection status of MNCs and the reducibility of the reaction system. With diverse combinations of surface protection status and reducibility, the final size of MNCs changes from 6.8 nm to 37.4 nm, and the morphology of the MNCs transforms from triangular prismatic to spherical (or quasi spherical), which indicates that the formation of MNCs with distinct sizes and morphologies is attributed to experiencing different pathways from nucleation to growth. Moreover, the reaction system is insensitive to the reaction temperature (220–240 °C) and time (2–6 hours), exhibiting a preferable operation error tolerance range for industrial application. Accordingly, a large scale synthesis is applied for synthesizing high quality MNCs, and 6 grams of spherical and monodispersed MNCs with a diameter of 11.2 ± 0.8 nm are obtained in a single reaction.

 Please wait while we load your content...
Please wait while we load your content...