Morphological control and upconversion luminescence of hollow CeO2 and Er3+–Yb3+ codoped CeO2 particles†
Abstract
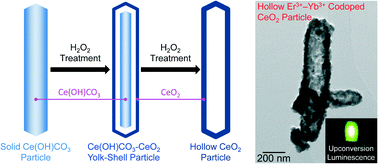
Hollow CeO2 particles with three types of morphologies (rod, rhombus, and dumbbell) were synthesized by retention of the morphology of Ce(OH)CO3 precursors during interfacial reactions of Ce(OH)CO3 with aqueous hydrogen peroxide solution. The wall areas of the hollow CeO2 particles comprised crystalline CeO2 nanoparticles. The pH of the aqueous hydrogen peroxide solution played a crucial role in the formation of the hollow structure. The mechanism underlying formation of the hollow structure was also evaluated. The hollow structures of the CeO2 particles with rod-shaped morphology were maintained even after calcination at 1000 °C, although the structures collapsed after calcination at 1250 °C. Similar reactions using CePO4 nanorods and plate-shaped Ce2(CO3)3·8H2O particles instead of Ce(OH)CO3 particles demonstrated that the plate-shaped Ce2(CO3)3·8H2O particles could be transformed into plate-shaped CeO2 particles, although the CePO4 nanorods were non-reactive. CeO2 rattle particles composed of CeO2 porous wall and dense CeO2 rod were also synthesized. Furthermore, by utilizing the excellent thermal stability of the hollow CeO2 particles with rod-shaped morphology, hollow Er3+–Yb3+ codoped CeO2 rods that display upconversion luminescence were synthesized by hydrogen peroxide treatment and subsequent calcination of Er3+–Yb3+ codoped Ce(OH)CO3 rods at 1000 °C.

 Please wait while we load your content...
Please wait while we load your content...