Polymer-fabricated synthesis of cerium oxide nanoparticles and applications as a green catalyst towards multicomponent transformation with size-dependent activity studies†
Abstract
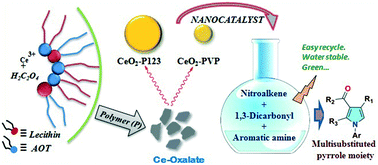
The present study primarily emphasizes the polymer-directed synthesis of cerium oxide nanoparticles (CeONP). Prior to the synthesis of CeONP, nanoflakes of the precursor cerium oxalate (CeOX) were first prepared by employing an AOT/lecithin/iso-octane/H2O mixed reverse microemulsion system and a polymer was added to the reaction medium to accomplish a dual purpose as both an additive and a structure-directing agent. Three polymers, namely, P123 (triblock copolymer), 17R4 (reverse triblock copolymer) and polyvinylpyrrolidone (PVP), were used as additives. Interestingly, the use of various polymers caused the synthesis of spherical cerium oxide nanoparticles within different size ranges (≈28.56 nm, 24.33 nm and 14.28 nm). Characterization of the synthesized CeONP was conducted by field emission scanning electron microscopy (FESEM), high-resolution transmission electron microscopy (HRTEM), X-ray diffraction spectroscopy (XRD) and Raman spectroscopy. The latter part of the study investigated their size-dependent catalytic activity in three-component reactions between nitrostyrene, 1,3-dicarbonyl compound, and aromatic primary amines to produce N-arylpyrrole as the desired product. The surface area of nanoceria (CeO2) was determined by BET measurements. Variations in size were found to directly affect the yield of the reactions. TEM analysis both before and after catalysis indicated the participation of the edges of the particles in the reaction.

 Please wait while we load your content...
Please wait while we load your content...