Inverted molecular cups: 1-D and 2-D Ag(i) coordination polymers from resorcinarene bis-thiacrowns†
Abstract
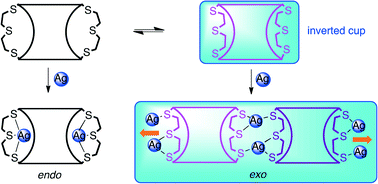
Resorcinarene bis-thiacrown hosts 1–3 were prepared and crystallized with silver trifluoroacetate yielding one and two dimensional Ag coordination polymers. The complexation of silver in exo-cavity fashion folds the thiacrown bridges inwards transforming the resorcinarene hosts into inverted molecular cups. The silver cations were coordinated to the resorcinarene ligands and trifluoroacetate anions, which act as monodentate or bidendate bridging ligands between the metal ions. Argentophilic Ag⋯Ag (2.93–3.38 Å) interactions supported by two bridging carboxylate anions were found in two of the structures, whereas longer Ag⋯Ag distances were observed if only one anion connected the silver cations. Both the anion and the resorcinarene lower rim alkyl chain had a role in the crystal packing, as indicated by the different metal coordination and packing tendencies of C-ethyl and C-pentyl bis-thiacrowns. A typical packing motif observed was a polymer chain with end-to-end joints and two Ag ions with tetrahedral coordination geometries. Solvent channels or discrete solvent pockets between the polymer layers were observed in all structures.
- This article is part of the themed collection: Form and Function of Molecular Cups and Capsules

 Please wait while we load your content...
Please wait while we load your content...